Fact Sheet: A Timeline of the Development of Fetal Pain Sensation
A previous fact sheet on the science of fetal pain was originally published in September 2022. This fact sheet has been substantially updated and organized according to a timeline of gestational development.
A 2007 manuscript on fetal pain notes:
It would be a mistake to view the onset of pain perception, or any other neural function, as we view assembly of computer parts. There is no scientific evidence that function in the multi-layered networks underlying pain perception wait for some cue to be “turned on.” The developing neural elements may be immature, but they are NOT inactive. … The immature pain system thus plays a signaling role during each stage of development and may use the neural elements available at that time to fulfill this role.[1]
This logical observation – that pain sensitivity, like other systems in the developing human, progressively matures rather than exhibiting all-or-none behavior – is critically important context in the face of historical assertions regarding fetal pain.
In adults, pain receptors convert noxious stimuli into neural signals sent via the spinal cord through the brainstem and into the thalamus, which relays them for processing in the cerebral cortex. In the developing fetus, early versions of many of these structures have been identified and shown to be active. For example, the mature cortex is preceded by a cortical plate and a transient structure called the subplate, which in turn develops from the transient preplate. Some groups have recently argued that early neural connections into this subplate are likely sufficient to transmit pain information, as discussed below. Fig. 1 provides a depiction of these developing pathways.[2]
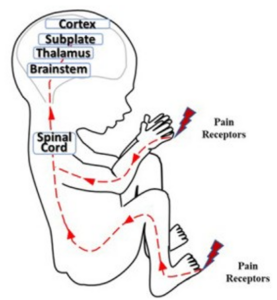
Fig. 1. Neural pathways for pain perception. Adapted from Thill.
This timeline describes key milestones in the development of pain perception, highlighting in particular recent evidence that fetuses have the capacity to experience pain by at least 12 to 15 weeks’ gestation.
Before 12 weeks’ gestation[3]
- The groundwork is laid for the circuitry responsible for sensing and responding to noxious stimuli (nociception). The first requirement for these circuits is sensory receptors which connect to the spinal cord and mediate a reflex response.[4] The embryo reflexively responds to touch around the mouth at 7-8 weeks of gestation, followed by much of the face as well as the palms, soles, and initial parts of the arm by weeks 10-11.[5]
- Substance P and enkephalins are two signaling molecules in the nervous system, also known as neurotransmitters, dedicated to processing pain. These two pain-specific neurotransmitters have been detected as early as week seven of gestation and increase in the following weeks.[6]
- Neural projections from the developing thalamus towards higher cortical structures are identified as early as nine weeks’ gestation.[7] These projections extend towards, and connect into, the developing subplate (also termed the pre-subplate at this stage) at 10-12 weeks.[8] As described below, this region of the brain and connections to it have been central to recent conclusions regarding fetal pain capacity.
12 weeks’ gestation
- In a recent review, Derbyshire and Bockmann note that “the evidence, and a balanced reading of that evidence, points towards an immediate and unreflective pain experience mediated by the developing function of the nervous system from as early as 12 weeks.”[9] This conclusion is in part drawn from the presence of the aforementioned neural projections into the subplate, about which the authors write: “There is now good evidence that [these] projections … are functional and equivalent to thalamocortical projections that emerge around 24 weeks’ gestation.”
-
- This work is particularly noteworthy given that Dr. Derbyshire was previously an author of several works, including the Royal College of Obstetricians and Gynaecologists (RCOG) report, which claimed that fetuses could not feel pain until at least 24 weeks’ gestation.[10]
- Beginning around 12 weeks’ gestation, the centro-median nucleus of the thalamus begins to segregate from the rest of nuclei of the thalamus.[11] The centro-median nucleus of the thalamus receives nociceptive information and participates in general arousal.[12]
- Sensation and response to touch continues to develop, extending to the limbs and parts of the chest.[13]
- Though typically performed later in gestation, surgical fetal interventions have been performed as early as 12 weeks’ gestation.[14] A consensus document on fetal anesthesia notes that “because it remains uncertain exactly when a fetus has the capacity to feel pain, it is best to administer adequate fetal anesthesia in all invasive maternal-fetal procedures.”[15]
13-14 weeks’ gestation
- Intentionality and awareness are demonstrated in goal-oriented movements of the fetus, including slower and more deliberate movements of co-twins towards one another.[16] Such movements have been proposed as indicating “a basic minimum level of consciousness” which continues to develop along a continuum, akin to a dimmer switch, which is pertinent to considerations of the fetus’s ability to perceive pain.[17]
- Neural projections into the cortical subplate continue to develop: additional research groups identify projections from the thalamus at 14 weeks.[18]
- A recommendation has been made that fetal anesthesia for invasive procedures be administered from 14 weeks onward.[19]
- Sensation and response to touch continues to develop, extending to the remainder of the chest, back, sides of the trunk, and the tongue.[20]
15 weeks’ gestation
- A report summarizes that the brain stem, thalamus, and insula are developed such that “an early form of pain may appear from the 15th week of gestation onward … [which] is diffuse and can dominate the overall perception of the fetus.”[21]
- Sensation and response to touch continues to develop, extending to the abdomen.[22]
16-20 weeks’ gestation
- Stress responses such as evasive movements, changes in blood flow, increased levels of noradrenaline, β-endorphin, and cortisol, are demonstrated in response to noxious but necessary fetal procedures, such as injections, and incidental contact with the fetus during amniocentesis.[23]
- Surgeries performed on the fetus routinely involve separate anesthesia for the fetus and mother. This practice has gained widespread acceptance as it has been demonstrated that anesthetics given to the mother alone may not pass sufficiently to the fetus to guarantee fetal anesthesia.[24] Importantly, fetal hormonal stress responses are minimal or absent if proper anesthesia is used during invasive procedures, indicating a lack of pain response.[25]
21-24 weeks’ gestation
- Babies delivered preterm as early as 21 weeks’ gestation demonstrate reactions to painful stimuli which require active monitoring and management in NICUs.[26] These observations of neonates, and their treatment, directly refute the historical narrative that fetal pain is impossible until 24 weeks or later.[27]
- Monitoring of fetal procedures in utero continues to show stress responses which decrease after anesthesia is provided.[28] Facial expressions of pain in response to injections of anesthetic into the thigh are observed as early as 23 weeks.[29]
24 weeks’ gestation and later
- Connections between the thalamus and cortex are made beginning at 24 weeks.[30] These connections are the final stage in the circuitry used throughout the remainder of life for pain processing and perception. While prior assertions regarding the limits of fetal pain perception used this as their basis,[31] more recent reviews of evidence have concluded that the capacity to experience pain does not require these connections.[32]
Additional notes on fetal pain
Heightened intensity of fetal pain
- Because inhibitory pathways “which are used to block painful stimuli … mature only after birth,” authors of one of the above-cited studies note that the fetus is more prone to pain than adults.[33] Additionally, premature infants display heightened brain responses to noxious stimuli compared to age-matched infants who were not born prematurely.[34] This heightened pain is also diffuse, as pain specificity maps mature with age.[35]
Further evidence refuting the necessity of the cortex in pain perception
- Scientific evidence apart from neuroembryology refutes the notion that connection to the cortex is required for pain perception: humans born without a cortex or who have part of their cortex removed still experience pain.[36]
- Further, while stimulation of higher cortical structures only in rare instances influences pain perception,[37] deep brain stimulation targeting early-developing subcortical structures is an effective therapy for managing chronic pain.[38]
The history of the 24-week assertion
- Based largely on studies from the early 2000s, professional societies asserted – and often currently maintain – that fetuses definitively cannot experience pain until at least the 24th week of gestation.[39] As noted above, subsequent reviews of the available scientific evidence have undermined this position, leading an author of one such report to publicly reverse his position.[40]
-
- Separate from the presence of more recent evidence refuting these premises, these conclusions were also criticized immediately after publication. A prominent British neonatologist writing about RCOG’s position noted: “We could rewrite this as ‘in theory they can’t feel pain, therefore they don’t.’”[41]
- These positions were in part based on the 1979 definition of pain by the International Association for the Study of Pain (IASP) as “an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage.”[42] This definition has been increasingly questioned as “limited and inadequate,” “demanding,” and “not of help” given that it “restricts pain almost exclusively to fairly mature human beings.”[43]
-
- The requirement of a sensory and emotional experience meant that “pain is no longer regarded as merely a physical sensation of noxious stimulus and disease, but is a conscious experience, modulated by mental, emotional and sensory mechanisms” and “an emergent property of social awareness.”[44] Before his public reversal in 2020, Dr. Derbyshire wrote that “by this definition, the neural tissue of the fetus at any stage of development cannot support pain experience.”[45]
-
- Another European group noted: “Whereas evidence for conscious pain perception is indirect, evidence for the subconscious incorporation of pain into neurological development and plasticity is incontrovertible.”[46]
-
- It is also noteworthy that the original 1979 definition was constructed at a time before even neonates were considered to possess the capacity for pain. A 1992 study randomly assigned adequate or inadequate anesthesia for four- to ten-day old neonates during cardiac surgery and found that the deeper anesthesia both reduced stress responses and resulted in fewer postoperative deaths.[47] Reflecting on this study, a separate author notes that “today, this study could not be ethically repeated.”[48] The evolving understanding of neonatal and fetal pain demands a more nuanced definition.
-
- Appreciating this nuance, Derbyshire and Bockmann draw a distinction in Reconsidering Fetal Pain between the “subjective reflection” required by the IASP definition and an “immediate and unreflective experience of pain” as early as 12 weeks. They then defend this early experience of pain as having both physiological and ethical importance.[49]
[1] Curtis L. Lowery et al., “Neurodevelopmental Changes of Fetal Pain,” Seminars in Perinatology, Pain, 31, no. 5 (October 1, 2007): 275–82, https://doi.org/10.1053/j.semperi.2007.07.004.
[2] Bridget Thill, “The Fetal Pain Paradox,” Frontiers in Pain Research 4 (March 21, 2023), https://doi.org/10.3389/fpain.2023.1128530.
[3] Throughout this work, points on the timeline are expressed in gestational age. The prenatal age methodology of reference material was adjusted accordingly as required.
[4] Brusseau R. Developmental Perspectives: Is the Fetus Conscious? International Anesthesiology Clinics 2008;46(3):11-23. doi:10.1097/aia.0b013e318181a88e.
[5] Tryphena Humphrey, “Some Correlations between the Appearance of Human Fetal Reflexes and the Development of the Nervous System,” in Progress in Brain Research, ed. Dominick P. Purpura and J. P. Schadé, vol. 4, Growth and Maturation of the Brain (Elsevier, 1964), 93–135, https://doi.org/10.1016/S0079-6123(08)61273-X.
[6] C. B Lou et al., “Localization of Substance P and Enkephalin by Immunohistochemistry in the Spinal Cord of Human Fetus,” Neuroscience 27, no. 3 (December 1, 1988): 989–93, https://doi.org/10.1016/0306-4522(88)90202-3.
[7] Ayman Alzu’bi et al., “Thalamocortical Afferents Innervate the Cortical Subplate Much Earlier in Development in Primate than in Rodent,” Cerebral Cortex 29, no. 4 (April 1, 2019): 1706–18, https://doi.org/10.1093/cercor/bhy327.
[8] Alzu’bi et al.; Irina Bystron, Colin Blakemore, and Pasko Rakic, “Development of the Human Cerebral Cortex: Boulder Committee Revisited,” Nature Reviews Neuroscience 9, no. 2 (February 2008): 110–22, https://doi.org/10.1038/nrn2252.
[9] Stuart WG Derbyshire and John C. Bockmann, “Reconsidering Fetal Pain,” Journal of Medical Ethics 46, no. 1 (January 1, 2020): 3–6, https://doi.org/10.1136/medethics-2019-105701.
[10] Stuart W G Derbyshire, “Can Fetuses Feel Pain?,” BMJ : British Medical Journal 332, no. 7546 (April 15, 2006): 909–12; Stuart W.G Derbyshire, “Fetal Pain: Do We Know Enough to Do the Right Thing?,” Reproductive Health Matters 16, no. sup31 (January 2008): 117–26, https://doi.org/10.1016/S0968-8080(08)31370-6; Royal College of Obstetricians and Gynaecologists, “Fetal Awareness: Review of Research and Recommendations for Practice,” March 2010.
[11] Mojsilović, J., & Zečević, N. (1991). Early development of the human thalamus: Golgi and Nissl study. Early human development, 27(1-2), 119-144. https://doi.org/10.1016/0378-3782(91)90033-Y.
[12] Ilyas A, Pizarro D, Romeo AK, Riley KO, Pati S. The centromedian nucleus: Anatomy, physiology, and clinical implications. J Clin Neurosci. 2019 May; 63:1-7. doi: 10.1016/j.jocn.2019.01.050.
[13] Humphrey, “Some Correlations between the Appearance of Human Fetal Reflexes and the Development of the Nervous System.”
[14] Thomas Kohl et al., “Vesico-Amniotic Shunt Insertion Prior to the Completion of 16 Weeks Results in Improved Preservation of Renal Function in Surviving Fetuses with Isolated Severe Lower Urinary Tract Obstruction (LUTO),” Journal of Pediatric Urology 18, no. 2 (April 1, 2022): 116–26, https://doi.org/10.1016/j.jpurol.2022.01.002.
[15] Debnath Chatterjee et al., “Anesthesia for Maternal-Fetal Interventions: A Consensus Statement From the American Society of Anesthesiologists Committees on Obstetric and Pediatric Anesthesiology and the North American Fetal Therapy Network,” Anesthesia and Analgesia 132, no. 4 (April 1, 2021): 1164–73, https://doi.org/10.1213/ANE.0000000000005177.
[16] Asim Kurjak et al., “Fetal Hand Movements and Facial Expression in Normal Pregnancy Studied by Four-Dimensional Sonography,” Journal of Perinatal Medicine 31, no. 6 (2003): 496–508, https://doi.org/10.1515/JPM.2003.076; Aida Salihagic Kadic and Asim Kurjak, “Cognitive Functions of the Fetus,” Ultraschall in Der Medizin (Stuttgart, Germany: 1980) 39, no. 2 (April 2018): 181–89, https://doi.org/10.1055/s-0043-123469; Umberto Castiello et al., “Wired to Be Social: The Ontogeny of Human Interaction,” ed. Aldo Rustichini, PLoS ONE 5, no. 10 (October 7, 2010): e13199, https://doi.org/10.1371/journal.pone.0013199.
[17] Thill, “The Fetal Pain Paradox.”
[18] Željka Krsnik et al., “Growth of Thalamocortical Fibers to the Somatosensory Cortex in the Human Fetal Brain,” Frontiers in Neuroscience 11 (2017): 233, https://doi.org/10.3389/fnins.2017.00233; Mijna Hadders-Algra, “Early Human Brain Development: Starring the Subplate,” Neuroscience and Biobehavioral Reviews 92 (September 2018): 276–90, https://doi.org/10.1016/j.neubiorev.2018.06.017; Duque, A., Krsnik, Z., Kostovic, I., and Rakic, P. (2016). Secondary expansion of the transient subplate zone in the developing cerebrum of human and nonhuman primates. Proc. Natl. Acad. Sci. U.S.A. 113, 9892–9897. doi: 10.1073/pnas.1610078113
[19] Ritu Gupta, Ruwan Wimalasundera, and Phil Moore, “Anaesthesic Considerations in Fetal Therapy,” in Anaesthesia for Uncommon and Emerging Procedures, ed. Basavana G. Goudra, Preet Mohinder Singh, and Michael S. Green (Cham: Springer International Publishing, 2021), 277–88, https://doi.org/10.1007/978-3-030-64739-1_28.
[20] Humphrey, “Some Correlations between the Appearance of Human Fetal Reflexes and the Development of the Nervous System.”
[21] Slobodan Sekulic et al., “Appearance of Fetal Pain Could Be Associated with Maturation of the Mesodiencephalic Structures,” Journal of Pain Research 9 (2016): 1031–38, https://doi.org/10.2147/JPR.S117959.
[22] Humphrey, “Some Correlations between the Appearance of Human Fetal Reflexes and the Development of the Nervous System.”
[23] X. Giannakoulopoulos et al., “Fetal Plasma Cortisol and Beta-Endorphin Response to Intrauterine Needling,” Lancet (London, England) 344, no. 8915 (July 9, 1994): 77–81, https://doi.org/10.1016/s0140-6736(94)91279-3; Jeronima Teixeira et al., “Fetal Haemodynamic Stress Response to Invasive Procedures,” The Lancet 347, no. 9001 (March 1996): 624, https://doi.org/10.1016/S0140-6736(96)91327-6; Xenophon Giannakoulopoulos et al., “Human Fetal and Maternal Noradrenaline Responses to Invasive Procedures,” Pediatric Research 45, no. 4 (April 1999): 494–99, https://doi.org/10.1203/00006450-199904010-00007; Rachel Gitau et al., “Fetal Hypothalamic-Pituitary-Adrenal Stress Responses to Invasive Procedures Are Independent of Maternal Responses,” The Journal of Clinical Endocrinology & Metabolism 86, no. 1 (2001); Boris M. Petrikovsky and Gary P. Kaplan, “Fetal Responses to Inadvertent Contact with the Needle during Amniocentesis,” Fetal Diagnosis and Therapy 10, no. 2 (1995): 83–85, https://doi.org/10.1159/000264210.
[24] Carlo V. Bellieni, “Analgesia for Fetal Pain during Prenatal Surgery: 10 Years of Progress,” Pediatric Research 89, no. 7 (May 2021): 1612–18, https://doi.org/10.1038/s41390-020-01170-2.
[25] Fisk NM, Gitau R, Teixeira JM, Giannakoulopoulos X, Cameron AD, Glover VA. Effect of direct fetal opioid analgesia on fetal hormonal and hemodynamic stress response to intrauterine needling. Anesthesiology. (2001) 95(4):828–35. doi: 10.1097/00000542-200110000-00008.
[26] Sharyn Gibbins et al., “Pain Behaviours in Extremely Low Gestational Age Infants,” Early Human Development 84, no. 7 (July 2008): 451–58, https://doi.org/10.1016/j.earlhumdev.2007.12.007; Robin Pierucci, “Fetal Pain, Following the Evidence,” Charlotte Lozier Institute, December 21, 2021, https://lozierinstitute.org/fetal-pain-following-the-evidence/; Thill, “The Fetal Pain Paradox.”
[27] Thill, “The Fetal Pain Paradox”; Pierucci, “Fetal Pain, Following the Evidence.”
[28] N. M. Fisk et al., “Effect of Direct Fetal Opioid Analgesia on Fetal Hormonal and Hemodynamic Stress Response to Intrauterine Needling,” Anesthesiology 95, no. 4 (October 2001): 828–35, https://doi.org/10.1097/00000542-200110000-00008.
[29] Lisandra S. Bernardes et al., “Sorting Pain out of Salience: Assessment of Pain Facial Expressions in the Human Fetus,” Pain Reports 6, no. 1 (January 27, 2021): e882, https://doi.org/10.1097/PR9.0000000000000882; L. S. Bernardes et al., “Facial Expressions of Acute Pain in 23-Week Fetus,” Ultrasound in Obstetrics & Gynecology: The Official Journal of the International Society of Ultrasound in Obstetrics and Gynecology 59, no. 3 (March 2022): 394–95, https://doi.org/10.1002/uog.23709.
[30] Ivica Kostović and Milos Judas, “The Development of the Subplate and Thalamocortical Connections in the Human Foetal Brain,” Acta Paediatrica (Oslo, Norway: 1992) 99, no. 8 (August 2010): 1119–27, https://doi.org/10.1111/j.1651-2227.2010.01811.x.
[31] Royal College of Obstetricians and Gynaecologists. RCOG Fetal Awareness Evidence Review (2022). Available from: https://www.rcog.org.uk/guidance/browse-all-guidance/other-guidelines-and-reports/fetal-awareness-updated-review-of-research-and-recommendations-for-practice/ (Accessed January 11, 2025).
[32] Derbyshire and Bockmann, “Reconsidering Fetal Pain”; Sekulic et al., “Appearance of Fetal Pain Could Be Associated with Maturation of the Mesodiencephalic Structures”; Bridget Thill, “Fetal Pain in the First Trimester,” The Linacre Quarterly 89, no. 1 (February 2022): 73–100, https://doi.org/10.1177/00243639211059245.
[33] Sekulic et al., “Appearance of Fetal Pain Could Be Associated with Maturation of the Mesodiencephalic Structures.”
[34] Slater R, Fabrizi L, Worley A, Meek J, Boyd S, Fitzgerald M. Premature infants display increased noxious-evoked neuronal activity in the brain compared to healthy age-matched term-born infants. Neuroimage. (2010) 52(2):583–9. doi: 10.1016/j.neuroimage.2010.04.253.
[35] Laura Jones et al., “Widespread Nociceptive Maps in the Human Neonatal Somatosensory Cortex,” eLife 11 (2022): e71655, https://doi.org/10.7554/eLife.71655.
[36] Bjorn Merker, “Consciousness without a Cerebral Cortex: A Challenge for Neuroscience and Medicine,” The Behavioral and Brain Sciences 30, no. 1 (February 2007): 63–81; discussion 81-134, https://doi.org/10.1017/S0140525X07000891; Barb Aleman and Bjorn Merker, “Consciousness without Cortex: A Hydranencephaly Family Survey,” Acta Paediatrica 103, no. 10 (2014): 1057–65, https://doi.org/10.1111/apa.12718.
[37] Laure Mazzola et al., “Stimulation of the Human Cortex and the Experience of Pain: Wilder Penfield’s Observations Revisited,” Brain 135, no. 2 (February 1, 2012): 631–40, https://doi.org/10.1093/brain/awr265.
[38] Richard G. Bittar et al., “Deep Brain Stimulation for Pain Relief: A Meta-Analysis,” Journal of Clinical Neuroscience 12, no. 5 (June 1, 2005): 515–19, https://doi.org/10.1016/j.jocn.2004.10.005; Steven M. Falowski, “Deep Brain Stimulation for Chronic Pain,” Current Pain and Headache Reports 19, no. 7 (June 7, 2015): 27, https://doi.org/10.1007/s11916-015-0504-1.
[39] Royal College of Obstetricians and Gynaecologists, “Fetal Awareness: Review of Research and Recommendations for Practice”; American College of Obstetricians and Gynecologists, “Facts Are Important: Gestational Development and Capacity for Pain,” n.d., accessed January 3, 2025, https://www.acog.org/advocacy/facts-are-important/gestational-development-capacity-for-pain; Royal College of Obstetricians and Gynaecologists, “RCOG Fetal Awareness Evidence Review, December 2022,” December 2022, https://www.rcog.org.uk/media/gdtnncdk/rcog-fetal-awareness-evidence-review-dec-2022.pdf.
[40] Derbyshire and Bockmann, “Reconsidering Fetal Pain.”
[41] Martin Ward Platt, “Fetal Awareness and Fetal Pain: The Emperor’s New Clothes,” Archives of Disease in Childhood. Fetal and Neonatal Edition 96, no. 4 (July 2011): F236-237, https://doi.org/10.1136/adc.2010.195966.
[42] IASP Subcommittee on Taxonomy, “Pain Terms: A List with Definitions and Notes on Usage.,” Pain 6, no. 3 (June 1979): 249.
[43] M. E. Canepa, L. Raffini, and L. A. Ramenghi, “Terminology Matters: Is the International Association for the Study of Pain Definition of Pain Fully Satisfactory for Fetuses, Neonates, and Infants?,” Frontiers in Pain Research 5 (May 16, 2024), https://doi.org/10.3389/fpain.2024.1369945; Derbyshire and Bockmann, “Reconsidering Fetal Pain”; Carlo V. Bellieni, “New Insights into Fetal Pain,” Seminars in Fetal & Neonatal Medicine 24, no. 4 (August 2019): 101001, https://doi.org/10.1016/j.siny.2019.04.001.
[44] Derbyshire, “Fetal Pain.”
[45] Ibid.
[46] Curtis L. Lowery et al., “Neurodevelopmental Changes of Fetal Pain,” Seminars in Perinatology, Pain, 31, no. 5 (October 1, 2007): 275–82, https://doi.org/10.1053/j.semperi.2007.07.004.
[47] K.J.S. Anand and P.R. Hickey, “Halothane–Morphine Compared with High-Dose Sufentanil for Anesthesia and Postoperative Analgesia in Neonatal Cardiac Surgery,” New England Journal of Medicine 326, no. 1 (January 2, 1992): 1–9, https://doi.org/10.1056/NEJM199201023260101.
[48] Robin Pierucci, “Fetal Pain: The Science Behind Why It Is the Medical Standard of Care,” The Linacre Quarterly 87, no. 3 (August 2020): 311–16, https://doi.org/10.1177/0024363920924877.
[49] Derbyshire and Bockmann, “Reconsidering Fetal Pain.”