Update: COVID-19 Vaccine Candidates and Abortion-Derived Cell Lines
Updated June 2, 2021
Accurate information about the development and production of COVID-19 vaccines is essential, especially because many proposed candidates use newer molecular technologies for production of a viral vaccine. One concern regarding the ethical assessment of viral vaccine candidates is the potential use of abortion-derived cell lines in the development, production or testing of a vaccine. This analysis utilizes data from the primary scientific literature when available, along with data from clinical trial documents, reputable vaccine tracking websites, and published commercial information.1 It is the hope that by providing accurate data, recipients can make well-informed decisions regarding vaccine choices.
For additional background and guidance, please see:
* A Visual Aid to Viral Infection and Vaccine Production for a visual primer on the various strategies for viral vaccine production.
* COVID-19 Vaccines & Fetal Cell Lines for an infographic description of how fetal cell lines are sometimes used to produce vaccines.
Flow Chart for Creation and Testing of Vaccines
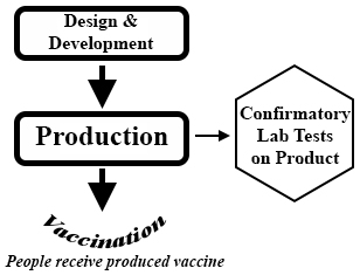 |
Design & Development: conceptualization, preparatory experiments, and specification for how vaccine will be constructed and produced.
Production: process used to manufacture final vaccine to be given to people.
Confirmatory Lab Tests on Product: tests to analyze quality, nucleic acid or protein sequence, protein conformation, antibody reactivity, etc. of final vaccine product.
Vaccination: giving final produced vaccine to people. |
||||||
To view this chart as a PDF, see: COVID-19 Vaccine Candidates and Abortion-Derived Cell Lines
DISCLAIMER: THIS WEBSITE DOES NOT PROVIDE MEDICAL ADVICE
The information, including but not limited to, text, graphics, images and other material contained on this website are for educational purposes only. No material on this site is intended to be a substitute for professional medical advice, diagnosis or treatment. Always seek the advice of your physician or other qualified health care provider with any questions you may have regarding a medical condition or treatment and before undertaking a new health care regimen, and never disregard professional medical advice or delay in seeking it because of something you have read on this website.