David Prentice, Ph.D.
Former Vice President and Director of Research for Charlotte Lozier InstituteDavid A. Prentice is a guest contributor for the Charlotte Lozier Institute. He is also Advisory Board Chair and a Founding Member for the Midwest Stem Cell Therapy Center, a unique comprehensive stem cell center in Kansas that he was instrumental in creating. In 2020, he was appointed by the Secretary of HHS to the federal Human Fetal Tissue Ethics Advisory Board. Dr. Prentice has almost 50 years’ experience as a scientific researcher and professor, including previous service as senior fellow for life sciences at the Family Research Council, Professor of Life Sciences at Indiana State University, Adjunct Professor of Medical and Molecular Genetics, Indiana University School of Medicine, and Adjunct Professor of Molecular Genetics at the John Paul II Institute, Catholic University of America.
He established Stem Cell Research Facts, an educational website providing scientific facts and patient-centered videos about adult stem cells, and is a founding member of Do No Harm: The Coalition of Americans for Research Ethics, and an advisory board member for the Center for Bioethics and Human Dignity. He has provided scientific advice for numerous medical and science professionals, legislators, policymakers and organizations at the state, federal, and international levels.
Dr. Prentice received his Ph.D. in biochemistry from the University of Kansas, and was at Los Alamos National Laboratory and the University of Texas Medical School-Houston before joining Indiana State University where in addition to his research and teaching, he served as Acting Associate Dean of Arts and Sciences and Assistant Chair of Life Sciences. He was recognized with the University’s Caleb Mills Distinguished Teaching Award and Faculty Distinguished Service Award. He has taught subjects ranging from non-majors biology to advanced and graduate courses including developmental biology, embryology, cell and tissue culture, history of biology, science and politics, pathophysiology, medical genetics, and medical biochemistry. Several of his courses were also taught on-line.
He received the 2007 Walter C. Randall Award in Biomedical Ethics from the American Physiological Society, given for promoting the honor and integrity of biomedical science through example and mentoring in the classroom and laboratory. He was honored in 2018 with selection for the 31st A. Kurt Weiss Lectureship on Biomedical Ethics, Oklahoma University Health Science Center. Dr. Prentice’s research interests encompass various aspects of cell growth control, cell and developmental biology; one major focus is adult stem cells. He has reviewed for various professional publications including The Journal of the American Medical Association.
He is an internationally-recognized expert on stem cell research, cell biology and bioethics, and has provided scientific lectures and policy briefings in 40 states and 21 countries, including testimony before the U.S. Congress and numerous state legislatures, the U.S. National Academy of Sciences, the U.S. President’s Council on Bioethics, European Parliament, British Parliament, Canadian Parliament, Australian Parliament, German Bundestag, French Senate, Swedish Parliament, the United Nations, and the Vatican. He was selected by President George W. Bush’s U.S. President’s Council on Bioethics to write the comprehensive review of adult stem cell research for the Council’s 2004 publication “Monitoring Stem Cell Research.”
Dr. Prentice has published numerous scientific and bioethics articles, including a review of stem cell science and adult stem cell treatments published in Circulation Research. He has also published numerous commentaries and op-eds, and travels nationally and internationally to give invited lectures regarding stem cell research, fetal tissue research, gene editing, cloning, embryology, cell and developmental biology, cell culture and vaccines, bioethics, and public policy. He has been interviewed in virtually all major electronic and print media outlets, including CNN, ABC, NBC, CBS, Fox, CSPAN, Reuters, AP, NPR, USA Today, BBC, The Washington Post, The Los Angeles Times, and The New York Times.
Research Authored
Stopping the Pain and Saving Lives: Successful Treatments for Sickle Cell Disease
The U.S. Food and Drug Administration (FDA) recently approved not just one, but two new gene therapies for sickle cell disease. The success of adult stem cell therapies continues to demonstrate that the progress of science and medicine need not rely on ethically compromised research and treatment approaches.
Charlotte Lozier Scholars Discuss Bioethical Challenges and Exploiting Nascent Human Beings
On Friday, September 22, 2023, Dr. David Prentice and Dr. Tara Sander Lee presented at Faith and Law's Friday Forum session on Capitol Hill.
 Tara Sander Lee, Ph.D.
Tara Sander Lee, Ph.D.
 David Prentice, Ph.D.
David Prentice, Ph.D.
CLI Comment: HHS Proposed Rule Fetal Tissue Regulations
The trafficking of aborted baby body parts for research is barbaric. The Trump administration was dedicated to ending this practice and protecting the sanctity of human life. Grave injustices were exposed that exploited unborn children for experimentation, but were not fully remedied.
 David Prentice, Ph.D.
David Prentice, Ph.D.
 Tara Sander Lee, Ph.D.
Tara Sander Lee, Ph.D.
Written Testimony of David A. Prentice, Ph.D. in Support of Prohibiting Prenatal Discrimination
On March 17, 2021, the Arizona House Judiciary Committee held a hearing on SB 1457, a bill to prohibit prenatal discrimination. Charlotte Lozier Institute’s Vice President and Research Director David A. Prentice, Ph.D., was invited by Senator Nancy Barto to testify as an expert witness, and submitted the following testimony.
What you need to know about the COVID-19 vaccines
CLI’s analysis found that a majority of vaccine candidates did not use abortion-derived cell lines in their production. Several used abortion-derived cell lines in laboratory testing, or their use in testing could not be determined. Experts urge all vaccine developers to use noncontroversial alternatives.
CLI Responds to Misinformation Regarding President Trump’s COVID-19 Treatment
“Uninformed commentary has emerged this morning stating that President Trump has received a medication created with the use of human embryonic stem cells. CLI has reviewed the published information and our conclusion is straightforward: The president was not given any medicines to treat COVID-19 that involved the destruction of human life. No human embryonic stem cells or human fetal tissue were used to produce the treatments President Trump received--period.
 David Prentice, Ph.D.
David Prentice, Ph.D.
 Tara Sander Lee, Ph.D.
Tara Sander Lee, Ph.D.
Update: COVID-19 Vaccine Candidates and Abortion-Derived Cell Lines
Accurate information about the development and production of COVID-19 vaccines is essential, especially because many proposed candidates use newer molecular technologies for production of a viral vaccine. One concern regarding the ethical assessment of viral vaccine candidates is the potential use of abortion-derived cell lines in the development, production or testing of a vaccine. This analysis utilizes data from the primary scientific literature when available, along with data from clinical trial documents, reputable vaccine tracking websites, and published commercial information.1 It is the hope that by providing accurate data, recipients can make well-informed decisions regarding vaccine choices.
Designing Babies: Science, Ethics, and Policies of a Post-Genomic Era
At the Center for Bioethics and Human Dignity annual summer conference "Bioethics in Real Life,” Dr. David Prentice and Dr. Tara Sander Lee presented a workshop entitled Designing Babies: Science, Ethics, and Policies of a Post-Genomic Era. Scientific advancements in the fields of genetics, molecular diagnosis, and genetic engineering were introduced so that participants could better understand the benefits, risks, limitations, policies, and potential impact on society. The ethics were carefully evaluated and discussed.
 Tara Sander Lee, Ph.D.
Tara Sander Lee, Ph.D.
 David Prentice, Ph.D.
David Prentice, Ph.D.
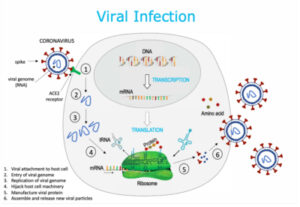
A Visual Aid to Viral Infection and Vaccine Production
As a result of the rapid spread of the new coronavirus technically known as SARS-CoV-2, the United States and other governments have begun crash efforts to create an effective vaccine or vaccines against the illness at “warp speed.” An array of candidates for this vaccine have emerged and are in various stages of development and testing. At the same time, questions have emerged about the ways in which these vaccines, reflecting experience across the history of development of these preventives, are produced, including processes to which millions of Americans object on moral grounds. The variety of means used to produce vaccines today is immense, employing both ethically licit and questionable procedures. To assist in understanding these modes of production, we offer this primer on the basic science involved.
 David Prentice, Ph.D.
David Prentice, Ph.D.
 Tara Sander Lee, Ph.D.
Tara Sander Lee, Ph.D.
Muestra de la infección viral y la producción de vacunas
Como resultado de la rápida propagación del nuevo coronavirus, técnicamente conocido como SARS-CoV-2, los gobiernos de Estados Unidos y de otros paises han pasado por varios intentos fallidos en la producción de una vacuna efectiva contra esta enfermedad que se transmite tan rápidamente. Ha surgido una selección de candidatos para esta vacuna y se encuentran en distintas etapas de desarrollo y de prueba. Al mismo tiempo, se han manifestado otras preguntas acerca de la manera en la cual estas vacunas son producidas; inlcuyendo los procesos sobre los cuales millones de americanos tienen objeciones en material moral. Hoy en día, la variedad de medios utilizados para producir vacunas es inmensa; en algunos casos son éticament lícitas, mientras que en otros los procedimientos que utilizan son questionables. Para ayudar a la comprensión de estos modos de producción, ofrecemos una guía básica respecto a la ciencia involucrada.
 David Prentice, Ph.D.
David Prentice, Ph.D.
 Tara Sander Lee, Ph.D.
Tara Sander Lee, Ph.D.