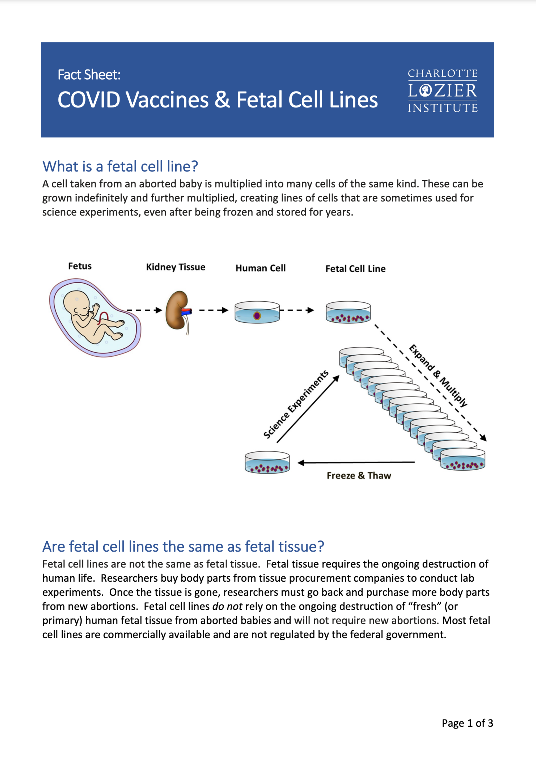
Most COVID-19 Vaccines Do Not Use Fetal Cells for Production; Several Used Abortion-Derived Cell Lines in Testing; Experts Urge All Vaccine Developers to Use Noncontroversial Alternatives
SHORT CHARTFULL CHART
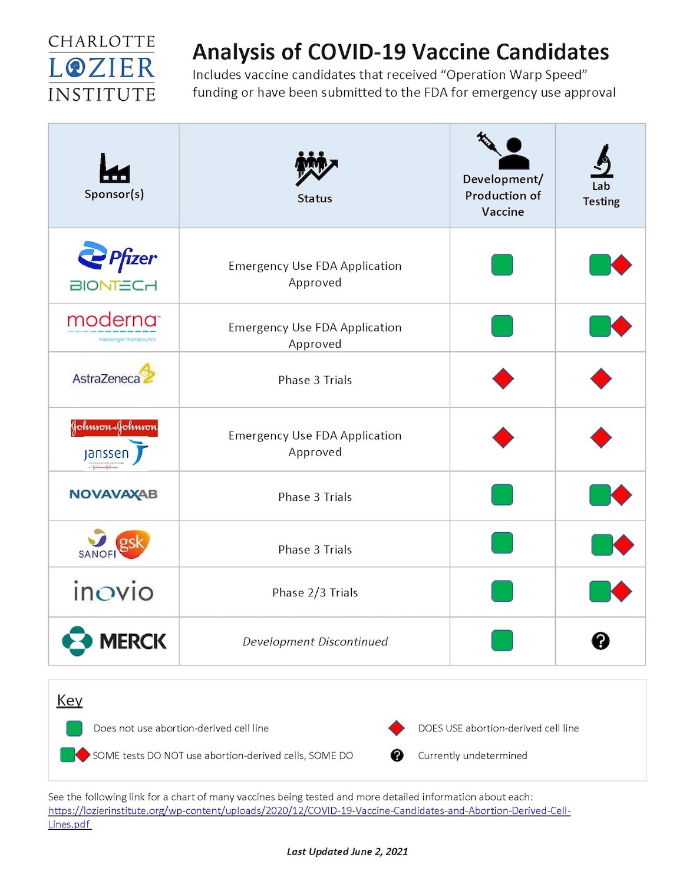
Charlotte Lozier Institute (CLI) has published a new chart (en español) detailing whether the eight leading COVID-19 vaccine candidates supported by Operation Warp Speed are produced or tested using cells derived from abortions. The vaccine candidates evaluated are those produced by Pfizer/BioNTech, Moderna, AstraZeneca, Johnson & Johnson, Novavax, Sanofi/GSK, Inovio, and Merck.
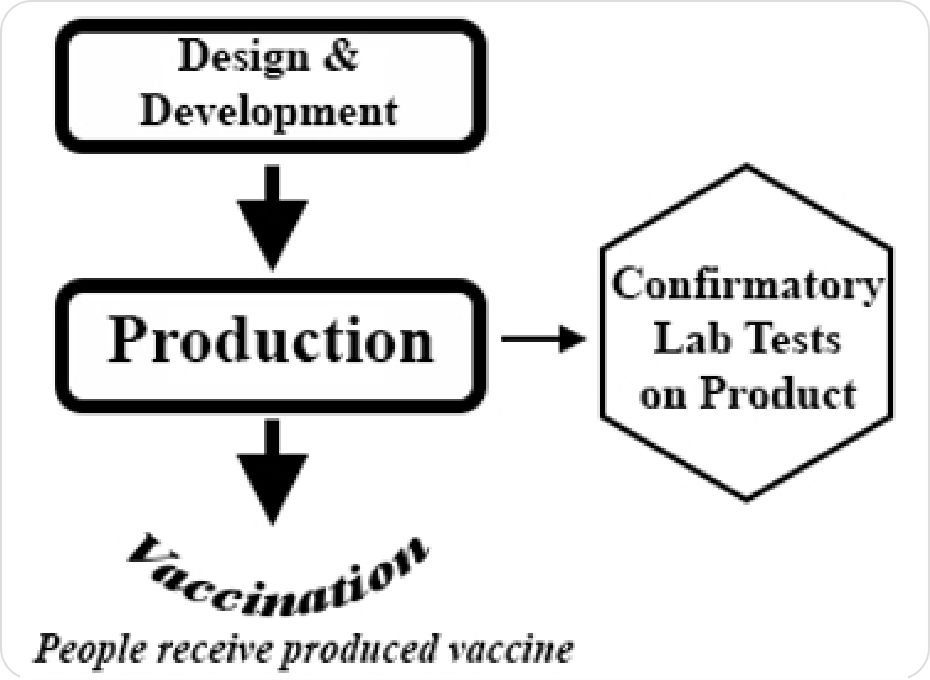
CLI’s analysis found that a majority of vaccine candidates did not use abortion-derived cell lines in their production. Several used abortion-derived cell lines in laboratory testing, or their use in testing could not be determined.
Accurate information about the development and production of COVID-19 vaccines is essential, especially because many proposed candidates use newer molecular technologies for production of a viral vaccine. CLI’s analysis utilizes data from the primary scientific literature when available, along with data from clinical trial documents, reputable vaccine tracking websites, and published commercial information.
It remains to be seen which vaccines will be approved for emergency use and how soon they will be available to patients. CLI will continue to provide the public with access to timely and accurate information so that individuals and families can weigh all factors – including secular science and religious traditions – and make vaccine decisions in line with their conscience.