A Visual Aid to Viral Infection and Vaccine Production
This is Issue 1 of the On Science.
Updated November 2020
Introduction
As a result of the rapid spread of the new coronavirus technically known as SARS-CoV-2, the United States and other governments have begun crash efforts to create an effective vaccine or vaccines against the illness at “warp speed.” An array of candidates for this vaccine have emerged and are in various stages of development and testing. At the same time, questions have emerged about the ways in which these vaccines, reflecting experience across the history of development of these preventives, are produced, including processes to which millions of Americans object on moral grounds. The variety of means used to produce vaccines today is immense, employing both ethically licit and questionable procedures. To assist in understanding these modes of production, we offer this primer on the basic science involved.
The Human Cell
From day 1 of egg fertilization and subsequent embryo development, every human being consists of a unique genetic code (or genome) consisting of deoxyribonucleic acid (or DNA). This genetic information is tightly packaged and arranged on chromosomes (numbered 1-22, plus 2 sex chromosomes X and/or Y) and stored in the nucleus of each human cell.
DNA consists of four different nucleotides (A, G, C, T) that join together on a single strand and make a sequence. DNA is normally found as a double stranded molecule, in which two separate DNA strands are wound around each other to form a double helix and each nucleotide of one strand forms a base pair with the nucleotide of the opposite strand.
In the same way that 26 letters of the alphabet combine to create a countless number of words and sentences for communicating, so it is with DNA. The four nucleotides combine in different ways to create various sequences, called genes, which are fundamental units of genetic information that provide specific instructions for a particular property or function within the cell.
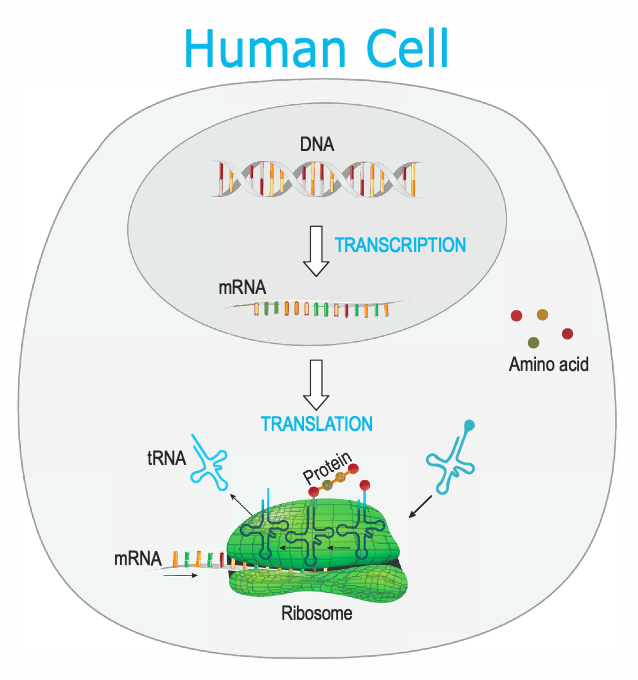
DNA is the original copy of genetic information, but DNA is not used as the direct source of instructions in the cell. Instead, DNA sends genetic information throughout the cell in the form of messenger ribonucleic acid (mRNA). mRNA is a nucleic acid sequence that is complementary to the template DNA strand, but slightly chemically different. The process by which DNA transfers information to mRNA is called transcription. As such, genes can be turned on and off, and messages sent and delivered throughout the cell, with exact precision.
Once mRNA is generated, the information it carries must be converted from one language to another. The mRNA “language” using nucleic acids, or genetic code, is translated to a protein “language” using amino acids. This mechanism of making protein using the information provided by mRNA is called translation. For every three nucleotide bases of mRNA (called a codon) that are translated, one amino acid is positioned in the protein sequence. The translated mRNA sequence will encode for a protein that consists of several amino acids strung together like pearls on a necklace. The final created protein product folds into a three-dimensional structure and carries out its specific function and purpose within the cell.
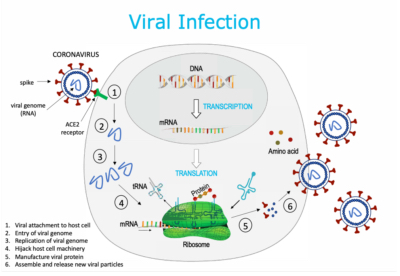
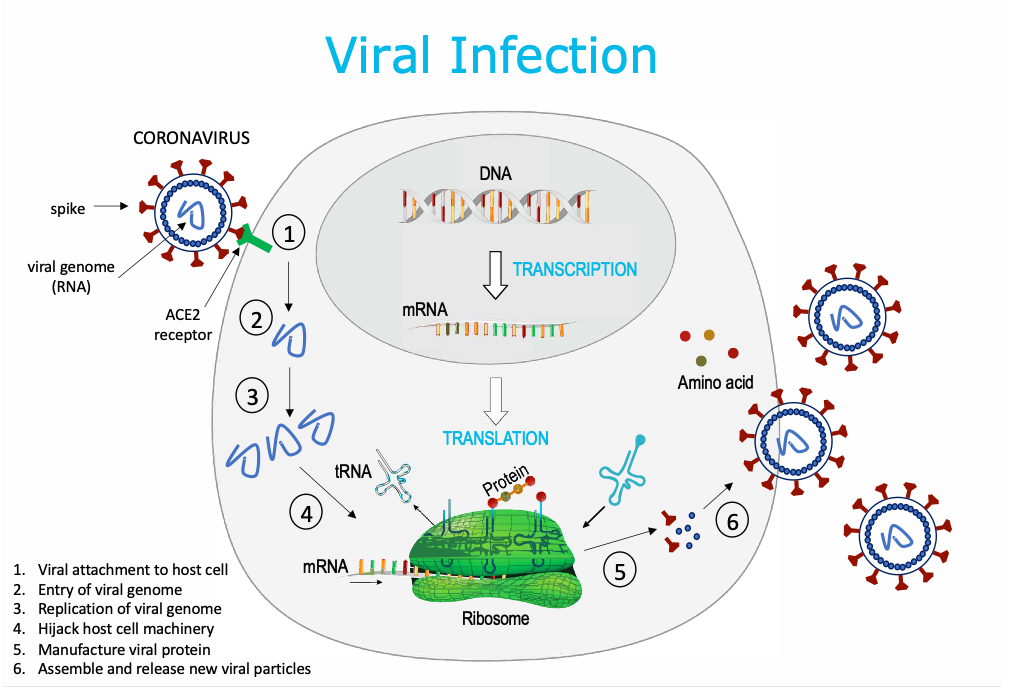
Viral Infection of the Human Cell
A virus infects the host and hijacks the cell machinery like a parasite in order to replicate (make more virus particles) and infect more host cells. The viral genome (DNA or RNA) enters a human cell, replicates more DNA and/or RNA sequence, and then uses the cell’s machinery to manufacture viral protein, so that more viral particles can be assembled and released into the host.
Coronaviruses (CoV) are a large family of single-stranded RNA viruses (about 30 kilobases in length) with a crown-like appearance due to the presence of spike surface glycoproteins on the envelope that surrounds the viral genome. These viruses can cross species barriers and cause, in humans, illness ranging from the common cold to more severe respiratory diseases such as Middle East Respiratory Syndrome (MERS-CoV) and Severe Acute Respiratory Syndrome (SARS-CoV). A new SARS-CoV-2 virus belonging to the coronavirus family causes a novel disease first identified in Wuhan, CHINA at the end of 2019, called COVID-19.
The first step of SARS-CoV-2 viral infection involves the binding of the spike protein on the coronavirus to the angiotensin-converting enzyme 2 (ACE2) entry receptor on the host cell. Once inside the host cell, the mRNA sequence of the SARS-CoV-2 genome will encode for several proteins including the spike surface glycoprotein. By this mechanism, the virus can produce more virulent particles for infecting other host cells.
Viral Vaccine Development
Five main strategies are being used to develop a vaccine against a newly recognized disease threat, the coronavirus SARS-CoV-2 that causes COVID-19. Several biotechnology companies, academic organizations and pharmaceutical companies are employing these different technologies in the race to bring their vaccine candidate to clinical trials.
The World Health Organization (WHO) hopes that a vaccine will be available by October 2021. Once developed with proven safety and efficacy, vaccines are injected into humans, to elicit an immune response including formation of antibodies as well as immune memory cells.
When a person is infected with the real pathogen, the immune response will remember the target and attack to prevent host cell infection. A brief description and simple illustration for each strategy is described below.
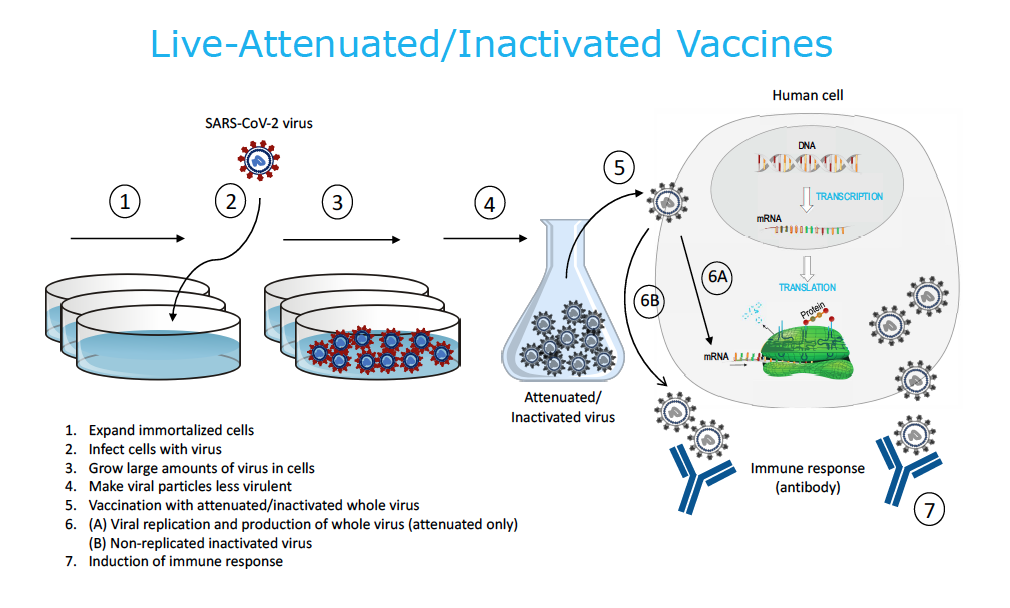
1. Live-attenuated/Inactivated Vaccines
As part of the manufacturing process, this type of vaccine is produced by growing large quantities of the whole virion in a controlled laboratory environment. The whole SARS-CoV-2 virus is “killed” (inactivated), “attenuated” (weakened), or a recoded version of the virus is synthesized that is less virulent. This technique requires cells to produce large quantities of the viral vaccine. Cultured human cell lines from aborted fetuses (e.g., PER.c6, HEK293) or ethical alternatives (e.g., K562, cord blood, placenta) are used. Animal cell lines are also available (e.g., Vero monkey cells, CHO hamster cells, Sf9 insect cells, etc.). Embryonated chicken eggs are also used for some vaccines. Once the vaccine is injected, the attenuated or inactivated viral vaccine replicates inside the cell, is released in the body and recognized as foreign by the immune system, and an immune response is generated. Live-attenuated/inactivated vaccines are infectious. There are existing, licensed human vaccines using this same platform.
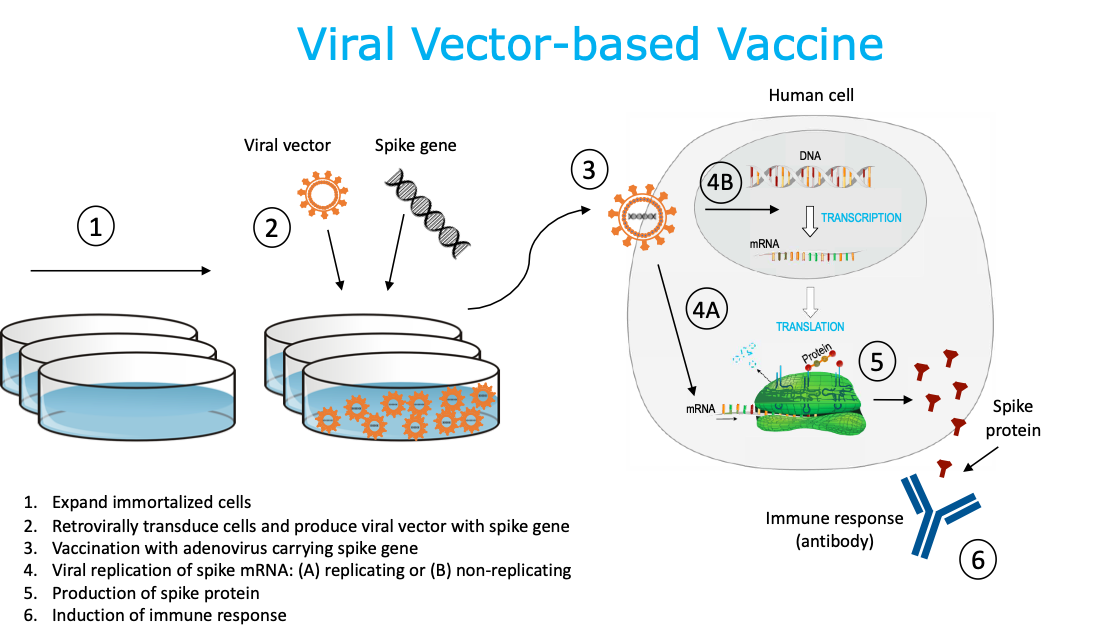
2. Viral Vector-based Vaccine
This technique utilizes a genetically engineered virus vector system (e.g., adenovirus, measles, VSV) that will carry a gene that encodes for a SARS-CoV-2 viral target, often the surface spike (S) protein. Cultured human cell lines (e.g., PER.c6, HEK293) are used like “factories” to produce large quantities of the engineered viruses. Alternatively, animal cell lines can also be used (e.g., Vero monkey cells, CHO hamster cells, Sf9 insect cells, etc.) Once injected, the viral vector-based vaccine enters the host cell and the engineered viral DNA is transcribed into mRNA using viral vector machinery (replicating) or machinery of the host cell (replication-deficient). The mRNA is then translated by the host cell machinery into SARS-CoV-2 spike protein, which is released from the cell. Pre-existing vector immunity might negatively affect vaccine effectiveness depending on the vector chosen (e.g., from previous adenovirus infection). Viral vector- based vaccines are non-infectious. There is an existing, licensed human vaccine using this same platform.
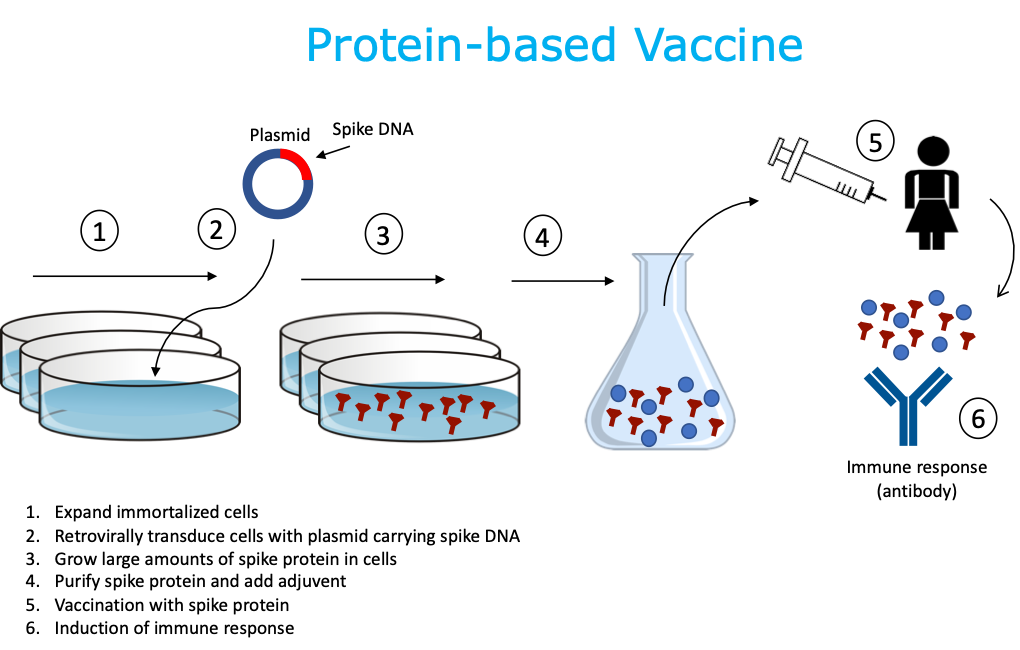
3. Protein-based Vaccine
The vaccine consists only of protein, such as the viral spike protein, which is synthesized and produced via recombinant genetics techniques in the lab, wherein a part of the protein (or subunit) is produced alone, without need for the whole, infectious growing pathogen. To make a recombinant or “synthetic” protein, DNA encoding for a region of the virus (e.g., spike) is combined with a plasmid that will help transport the DNA instructions for making viral protein. A host cell line is used to rapidly produce the protein. This protein is grown in bulk, collected, purified, combined with adjuvant to enhance the immune response, and then packaged as recombinant vaccine. Cultured human or animal cell lines are commonly used to produce the protein, as well as fertilized chicken eggs. Protein-based vaccines are non-infectious. There are existing, licensed human vaccines using this platform.
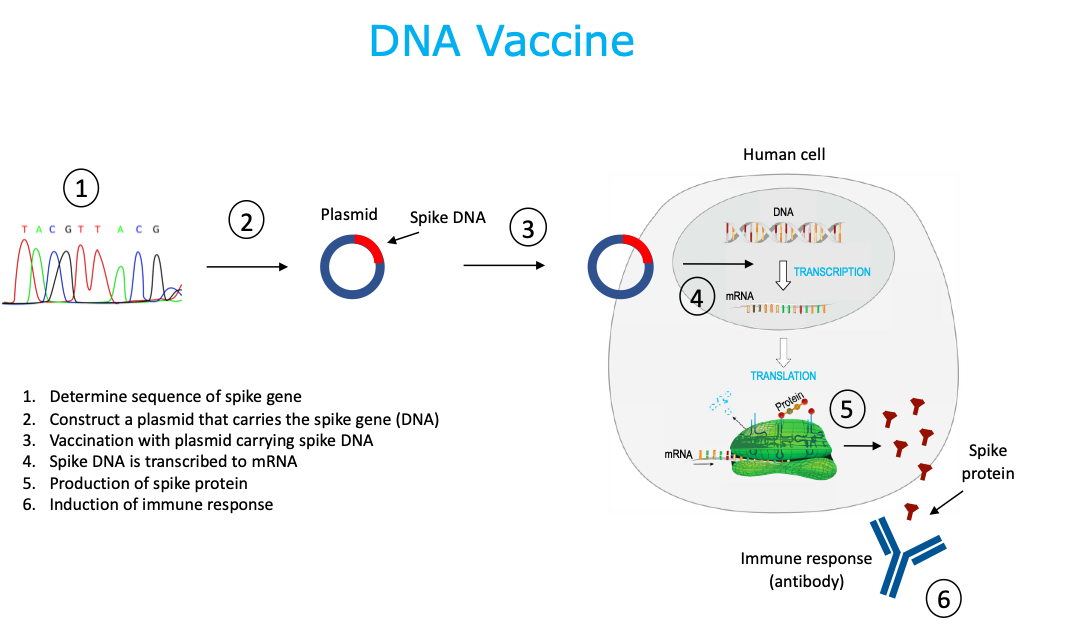
4. DNA Vaccine
A plasmid is synthesized that carries the spike gene that will encode for the SARS-CoV-2 spike protein. The DNA is chemically or enzymatically synthesized in the laboratory, without the use of cells. The plasmid enters the nucleus of the host cell. DNA is transcribed into mRNA, mRNA is translated to produce SARS-CoV-2 surface protein, and SARS-CoV-2 surface protein is released. DNA vaccines are non-infectious. This technology is new and there are currently no existing, licensed human vaccines using this platform.
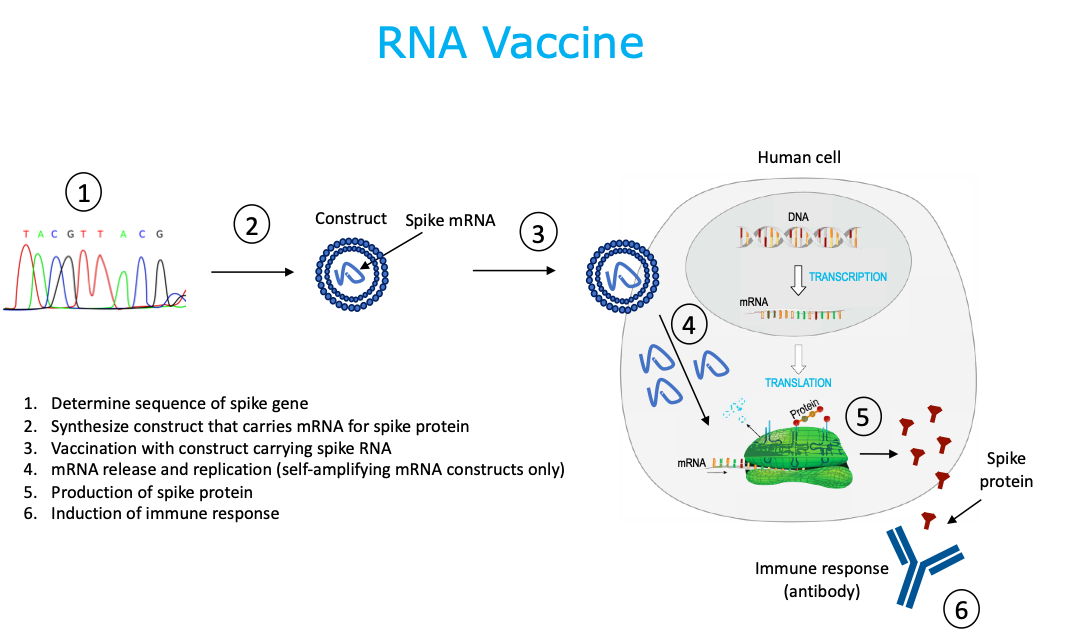
5. RNA Vaccine
RNA vaccines use mRNA that carries the code for the SARS-CoV-2 spike protein. These vaccines can be either non-replicating mRNA construct or self-amplifying mRNA construct, able to direct intracellular mRNA amplification. The mRNA is chemically or enzymatically synthesized in the laboratory, without the use of cells. Lipid nanoparticles encapsulate the mRNA constructs to protect them from degradation and promote cellular uptake. This is a cell-free system. mRNA vaccines are non-infectious. This technology is new and there are currently no existing, licensed human vaccines using this platform, but studies show they can elicit immunity against flu, Zika, rabies and coronavirus.
DISCLAIMER: THIS WEBSITE DOES NOT PROVIDE MEDICAL ADVICE
The information, including but not limited to, text, graphics, images and other material contained on this website are for educational purposes only. No material on this site is intended to be a substitute for professional medical advice, diagnosis or treatment. Always seek the advice of your physician or other qualified health care provider with any questions you may have regarding a medical condition or treatment and before undertaking a new health care regimen, and never disregard professional medical advice or delay in seeking it because of something you have read on this website.






























